*Data reflective of combined TYVASO DPI and nebulized TYVASO as of April 30, 2025.1
TYVASO DPI is dosed with just 1 breath per cartridge, 4x daily, approximately every 4 waking hours. Each dose is inhaled in less than 2 seconds.
The BREEZE study was designed to assess the safety and tolerability of switching from TYVASO to TYVASO DPI3
BREEZE Study Design: This open-label clinical study evaluated patients with PAH (N=51) who were on a stable dose of TYVASO and were switched to a corresponding dose of TYVASO DPI.3
Patients underwent safety and pharmacokinetic assessments and 6-minute walk tests, and also completed questionnaires about device preference and satisfaction.2
49 of 51 patients (96%) elected to continue therapy with TYVASO DPI in an OEP with follow-up visits every 8 weeks.2
Interim data demonstrated that patients could titrate higher than their initial dose of TYVASO DPI, with one patient titrating up to 176 mcg (~33 breaths), 4x daily.6
Select patient background information
-
Patients started TYVASO ≥3 months prior to the baseline visit and were on a stable regimen (no change in dose within 30 days of baseline visit) of TYVASO (6 to 12 breaths QID)3
-
80% of patients were on dual background therapy in addition to TYVASO3†
If receiving other approved background therapy, the patient was required to be on a stable dose with no additions or discontinuations for a minimum of 30 days3
-
61% of patients were FC II at screening3‡
-
FC IV patients were excluded from the study8
-
Patients had a mean baseline 6MWD of 418.9 m (SD 109.4)3
-
Patients had a forced expiratory volume in 1 second (FEV1) ≥60% and forced expiratory volume in 1 second/forced vital capacity (FEV1/FVC) ratio ≥60% during the 6 months prior to enrollment3
†84.3% endothelin receptor antagonist, 80.4% phosphodiesterase type 5 inhibitor, and 13.7% soluble guanylate cyclase stimulator.3
‡12% were FC I and 27% were FC III.3
Results
Adverse reactions: Consistent safety and tolerability when switching to TYVASO DPI2
During the 3-week treatment phase of the BREEZE study, most commonly reported (≥4% of patients) AEs in patients previously stable on TYVASO were2,3:
- Cough (35%)
- Headache (16%)
- Dyspnea (8%)
- Nausea (6%)
Discontinuation of TYVASO DPI due to an AE occurred in 2 (3.9%) patients.3
Patient tolerability, as assessed by incidence of new AEs following transition to TYVASO DPI, was consistent with the expected known safety profile of TYVASO.2

AE rate declined during the OEP, suggesting that TYVASO DPI tolerance may improve over time.2,3

Systemic exposure was similar between TYVASO DPI and TYVASO1,3
Mean treprostinil concentration over time (dose levels pooled)1
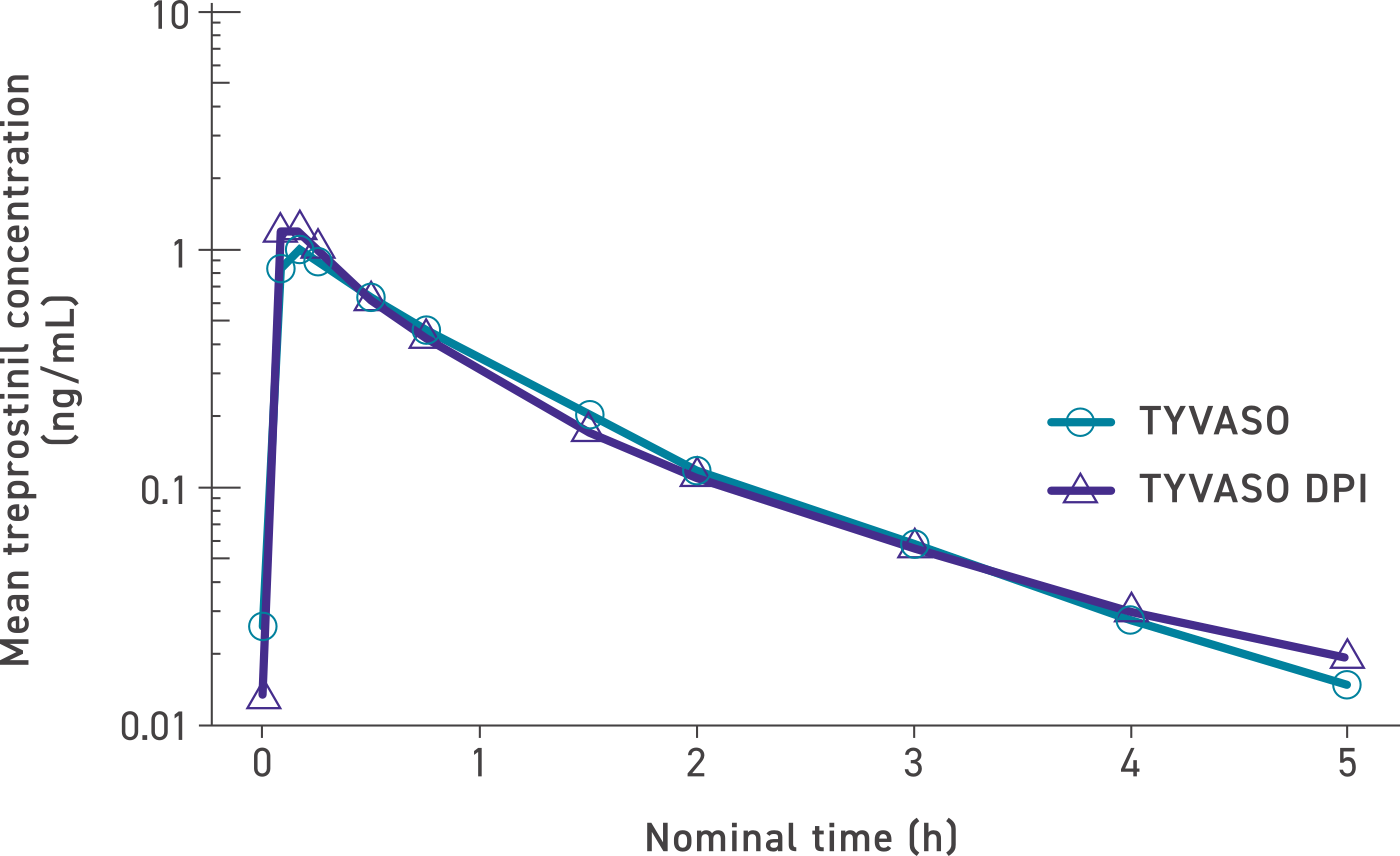
The maximum mean treprostinil concentration occurred rapidly for both TYVASO and TYVASO DPI in the low-, mid-, and high-dose groups: 32 mcg (6-7 breaths; n=2), 48 mcg
11.5 m additional increase in 6MWD (P=0.0217)3
For patients previously stable on TYVASO, TYVASO DPI demonstrated a significant improvement in 6MWD at week 3 compared with TYVASO treatment at baseline. Interim results of the OEP suggest that this improvement was sustained over 51 weeks.3

45/46 (P<0.0001)
REPORTED PATIENT SATISFACTION WITH TYVASO DPI
at week 3 compared with 31% (16/51) of patients satisfied with the TYVASO ultrasonic nebulizer at baseline.3


Learn more about TYVASO for the treatment of PAH and PH-ILD and the support available to you and your patients.9
TYVASO for PAH
TYVASO for PH-ILD






